
| Traditional endotoxin testing uses horseshoe crab blood — an ancient and irreplaceable marine resource |
| ACROBiosystems offers a recombinant Factor C (rFC) solution that’s not only accurate, but also sustainable. Let’s protect these ancient creatures while ensuring your quality control! |

| SAFENSURE™ Recombinant Factor C Endotoxin Detection Kit |
|
Assay Principles |
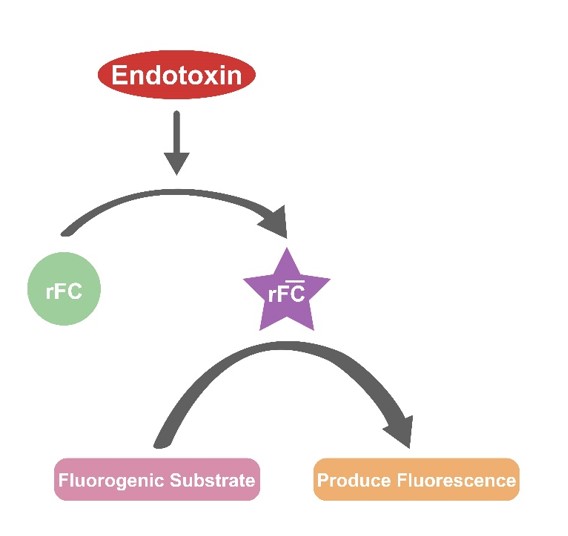
| The Recombinant Factor C Endotoxin Detection Kit uses recombinant technology to detect endotoxins by activating Factor C, which then cleaves a fluorogenic substrate to produce a measurable signal, as described in the original text. This fluorescent signal directly correlates with the endotoxin concentration, allowing for quantification on a white 96-well plate after a one-hour incubation at 37?. The measurement, conducted at 380/440 nm with a fluorescence reader, confirms contamination levels, as detailed further in. |
| Product Overview |
| The Recombinant Factor C Endotoxin Detection Kit is a novel endotoxin detection method based on the recombination technology. Recombinant Factor C, as the first component of the horseshoe crab coagulation cascade reaction, is activated by an endotoxin. The activated Factor C can cleave the fluorogenic substrate and produce a fluorescent signal. The increase of fluorescence signal is positively correlated with the dosage of endotoxin. The experiment is carried on a white 96-well plate and is measured at time zero and after a one-hour 37? incubation. Use a fluorescence microplate reader to measure at the wavelength of ex/em = 380/440 nm to determine whether the sample is contaminated by endotoxin. |
| Features, Advantages, Benefits |
|
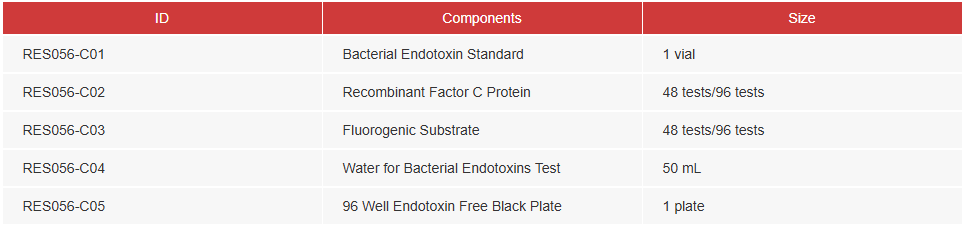
| Materials Provided |
 |

Come and meet us in Fisciano!